Lewis Dot Structure Covalent Bonds Calculator
- Lewis electron dot diagrams can be drawn to illustrate covalent bond formation. Double bonds or triple bonds between atoms may be necessary to 12.4: Covalent Bonds and Lewis Structures - Chemistry LibreTexts.
- The Na + cation and the Cl-anion are held together by electrostatic or ionic bonds.There is no sharing in ionic bonding. The anion takes the electron for itself and the cation is happy to get rid of its electron. The ions in ionic compounds are arranged in three-dimensional structures.
Here is the simple view on this important topic: In Lewis structures, we show each covalent bond as two dots which represent a pair of electrons. For example: Therefore, we can say that Lewis structures are electron dot representations for molecules. The trick here is to keep track of the electrons and correctly place the atoms in the molecule. How To Read A Lewis Dot Structure. The Lewis dot structure is drawn with letters that represent the atoms of the element, and then a number of dots or dashes surrounding these letters. Dots can be used to represent the shared electrons within the bonds of the atoms, but dashes can be used to represent covalent bonds as well. Electrons in the Lewis Dot Structure? ZDraw Lewis Structures for O 2 and N 2. 14 Covalent Bond zThere is an optimum distance between atoms in a covalent bond. This is the bond length: calculated by adding the radii of two atoms. Strengths of Covalent Bonds zCloser nuclei result in a stronger bond. Shorter bond = stronger bond.
Learning Objectives
- Define covalent bond.
- Illustrate covalent bond formation with Lewis electron dot diagrams.
Ionic bonding typically occurs when it is easy for one atom to lose one or more electrons, and for another atom to gain one or more electrons. However, some atoms will not give up or gain electrons easily. Yet they still participate in compound formation. How? There is another mechanism for obtaining a complete valence shell: sharing electrons. When electrons are shared between two atoms, they form a covalent bond.
Let us illustrate a covalent bond by using H atoms, with the understanding that H atoms need only two electrons to fill the 1s subshell. Each H atom starts with a single electron in its valence shell:
[mathbf{H, cdot }; ; ; ; ; mathbf{cdot : H}nonumber ]
The two H atoms can share their electrons:
[mathbf{H}: mathbf{: H}nonumber ]
We can use circles to show that each H atom has two electrons around the nucleus, completely filling each atom's valence shell:
Because each H atom has a filled valence shell, this bond is stable, and we have made a diatomic hydrogen molecule. (This explains why hydrogen is one of the diatomic elements.) For simplicity's sake, it is not unusual to represent the covalent bond with a dash, instead of with two dots:
H–H
Because two atoms are sharing one pair of electrons, this covalent bond is called a single bond. As another example, consider fluorine. F atoms have seven electrons in their valence shell:
These two atoms can do the same thing that the H atoms did; they share their unpaired electrons to make a covalent bond.
Note that each F atom has a complete octet around it now:
We can also write this using a dash to represent the shared electron pair:
There are two different types of electrons in the fluorine diatomic molecule. The bonding electron pair makes the covalent bond. Each F atom has three other pairs of electrons that do not participate in the bonding; they are called lone pair electrons. Each F atom has one bonding pair and three lone pairs of electrons.
Covalent bonds can be made between different elements as well. One example is HF. Each atom starts out with an odd number of electrons in its valence shell:

The two atoms can share their unpaired electrons to make a covalent bond:

Note that the H atom has a full valence shell with two electrons, while the F atom has a complete octet of electrons.
Example (PageIndex{1}):
Use Lewis electron dot diagrams to illustrate the covalent bond formation in HBr.
Solution
HBr is very similar to HF, except that it has Br instead of F. The atoms are as follows:
The two atoms can share their unpaired electron:
Exercise (PageIndex{1})
Use Lewis electron dot diagrams to illustrate the covalent bond formation in Cl2.
Answer
More than two atoms can participate in covalent bonding, although any given covalent bond will be between two atoms only. Consider H and O atoms:
Lewis Dot Structure Covalent Bonds
The H and O atoms can share an electron to form a covalent bond:
The H atom has a complete valence shell. However, the O atom has only seven electrons around it, which is not a complete octet. This can be fixed by including a second H atom, whose single electron will make a second covalent bond with the O atom:
(It does not matter what side the second H atom is positioned on.) Now the O atom has a complete octet around it, and each H atom has two electrons, filling its valence shell. This is how a water molecule, H2O, is made.
Example (PageIndex{2}):
Use a Lewis electron dot diagram to show the covalent bonding in NH3.
Solution
The N atom has the following Lewis electron dot diagram:

It has three unpaired electrons, each of which can make a covalent bond by sharing electrons with an H atom. The electron dot diagram of NH3 is as follows:
Exercise (PageIndex{2})
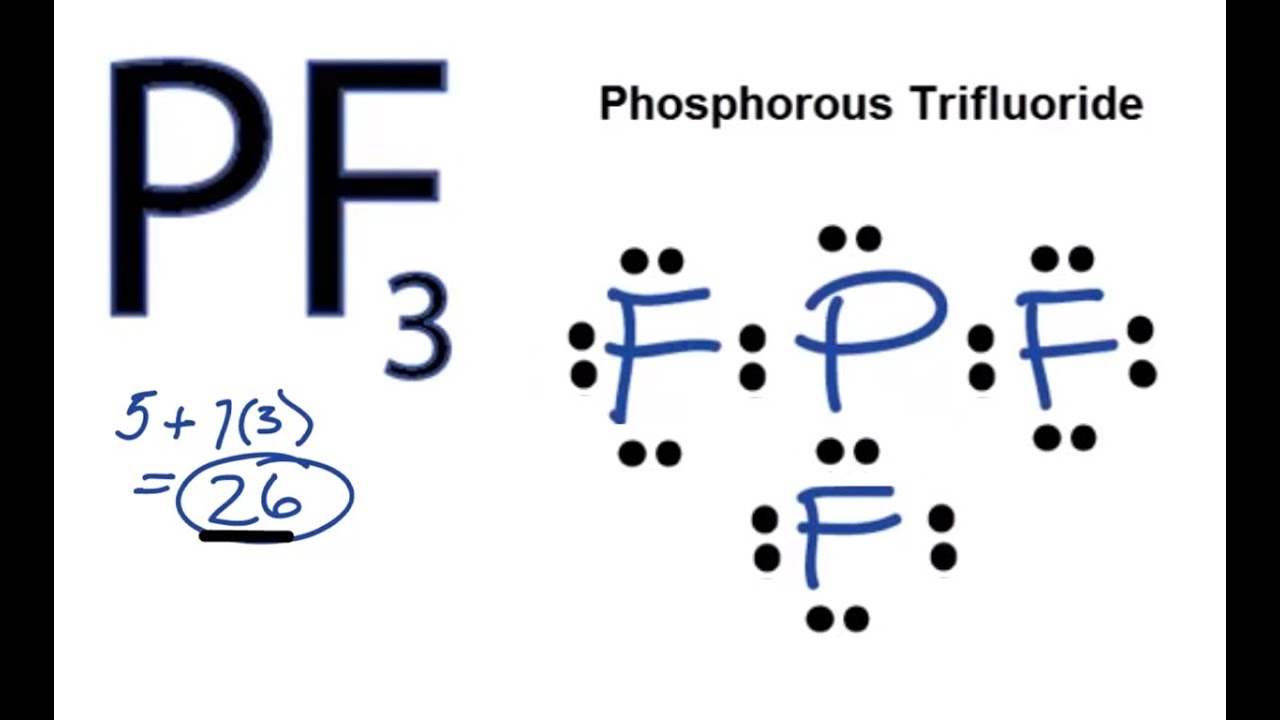
Use a Lewis electron dot diagram to show the covalent bonding in PCl3.
- Answer
There is a simple set of steps for determining the Lewis electron dot diagram of a simple molecule. First, you must identify the central atom and the surrounding atoms. The central atom is the atom in the center of the molecule, while the surrounding atoms are the atoms making bonds to the central atom. The central atom is usually written first in the formula of the compound (H2O is the notable exception). After the central and surrounding atoms have been identified, follow these steps:
- Count the total number of valence electrons. Add extra if the species has negative charges and remove some for every positive charge on the species.
- Write the central atom and surround it with the surrounding atoms.
- Put a pair of electrons between the central atom and each surrounding atom.
- Complete the octets around the surrounding atoms (except for H).
- Put remaining electrons, if any, around the central atom.
- Check that every atom has a full valence shell.
Let us try these steps to determine the electron dot diagram for BF4−. The B atom is the central atom, and the F atoms are the surrounding atoms. There is a negative sign on the species, so we have an extra electron to consider.
1. Count the total number of electrons. B has 3, each F has 7, and there is one extra electron: 3 + 7 + 7 + 7 + 7 + 1 = 32.
2. Write the central atom surrounded by surrounding atoms.
3. Put a pair of electrons between the central atom and each surrounding atom. This uses up eight electrons, so we have 32 − 8 = 24 electrons left.
4. Complete the octets around the surrounding atoms (except for H). This uses up 24 more electrons, leaving 24 − 24 = 0 electrons left.
5. Put remaining electrons, if any, around the central atom. There are no additional electrons to add to the central atom.
6. Check. The B atom has eight electrons around it, as does each F atom. Each atom has a complete octet. This is a good Lewis electron dot diagram for BF4−.
Sometimes, however, these steps do not work. If we were to follow these steps for the compound formaldehyde (CH2O), we would get the following:
The H and O atoms have the proper number of electrons, but the C atom only has six electrons around it, not the eight electrons for an octet. How do we fix this?
We fix this by recognizing that two atoms can share more than one pair of electrons. In the case of CH2O, the O and C atoms share two pairs of electrons, with the following Lewis electron dot diagram as a result:
By circling the electrons around each atom, we can now see that the O and C atoms have octets, while each H atom has two electrons:
Each valence shell is full, so this is an acceptable Lewis electron dot diagram. If we were to use lines to represent the bonds, we would use two lines between the C and O atoms:
The bond between the C and O atoms is a double bond and represents two bonding pairs of electrons between the atoms. If the rules for drawing Lewis electron dot diagrams do not work as written, a double bond may be required.
Example (PageIndex{3}): Carbon Dioxide
What is the proper Lewis electron dot diagram for CO2?
Solution
The central atom is a C atom, with O atoms as surrounding atoms. We have a total of 4 + 6 + 6 = 16 valence electrons. Following the rules for Lewis electron dot diagrams for compounds gives us
The O atoms have complete octets around them, but the C atom only has four electrons around it. The way to solve this dilemma is to make a double bond between carbon and each O atom:
Each O atom still has eight electrons around it, but now the C atom also has a complete octet. This is an acceptable Lewis electron dot diagram for CO2.
Exercise (PageIndex{3})
What is the proper Lewis electron dot diagram for carbonyl sulfide (COS)?
Answer
It is also possible to have a triple bond, in which there are three pairs of electrons between two atoms. Good examples of this are elemental nitrogen (N2) and acetylene (C2H2):
Acetylene is an interesting example of a molecule with two central atoms, which are both C atoms. Polyatomic ions are bonded together with covalent bonds. Because they are ions, however, they participate in ionic bonding with other ions. So both major types of bonding can occur at the same time.
Food and Drink App: Vitamins and Minerals
Vitamins are nutrients that our bodies need in small amounts but cannot synthesize; therefore, they must be obtained from the diet. The word vitamin comes from 'vital amine' because it was once thought that all these compounds had an amine group (NH2) in it. This is not actually true, but the name stuck anyway.
All vitamins are covalently bonded molecules. Most of them are commonly named with a letter, although all of them also have formal chemical names. Thus vitamin A is also called retinol, vitamin C is called ascorbic acid, and vitamin E is called tocopherol. There is no single vitamin B; there is a group of substances called the B complex vitamins that are all water soluble and participate in cell metabolism. If a diet is lacking in a vitamin, diseases such as scurvy or rickets develop. Luckily, all vitamins are available as supplements, so any dietary deficiency in a vitamin can be easily corrected.
A mineral is any chemical element other than carbon, hydrogen, oxygen, or nitrogen that is needed by the body. Minerals that the body needs in quantity include sodium, potassium, magnesium, calcium, phosphorus, sulfur, and chlorine. Essential minerals that the body needs in tiny quantities (so-called trace elements) include manganese, iron, cobalt, nickel, copper, zinc, molybdenum, selenium, and iodine. Minerals are also obtained from the diet. Interestingly, most minerals are consumed in ionic form, rather than as elements or from covalent molecules. Like vitamins, most minerals are available in pill form, so any deficiency can be compensated for by taking supplements.
Summary
- Covalent bonds are formed when atoms share electrons.
- Lewis electron dot diagrams can be drawn to illustrate covalent bond formation.
- Double bonds or triple bonds between atoms may be necessary to properly illustrate the bonding in some molecules.
Learning Objective
Lewis Dot Structure Covalent Bonds Calculator Formula
- Predict and draw the Lewis structure of simple covalent molecules and compounds
Key Points
- The octet rule says that the noble gas electronic configuration is a particularly favorable one that can be achieved through formation of electron pair bonds between atoms.
- In many atoms, not all of the electron pairs comprising the octet are shared between atoms. These unshared, non-bonding electrons are called ‘lone pairs’ of electrons.
- Although lone pairs are not directly involved in bond formation, they should always be shown in Lewis structures.
- There is a logical procedure that can be followed to draw the Lewis structure of a molecule or compound.
Terms
- Lewis structureFormalism used to show the structure of a molecule or compound, in which shared electrons pairs between atoms are indicated by dashes. Non-bonding, lone pairs of electrons must also be shown.
- covalent bondTwo atoms share valence electrons in order to achieve a noble gas electronic configuration.
- exceptions to the octet ruleHydrogen (H) and helium (He) only need two electrons to have a full valence level.
- octet ruleAtoms try to achieve the electronic configuration of the noble gas nearest to them in the periodic table by achieving a full valence level with eight electrons.
The Octet Rule
Noble gases like He, Ne, Ar, Kr, etc., are stable because their valence level is filled with as many electrons as possible. Eight electrons fill the valence level for all noble gases, except helium, which has two electrons in its full valence level. Other elements in the periodic table react to form bonds in which valence electrons are exchanged or shared in order to achieve a valence level which is filled, just like in the noble gases. We refer to this chemical tendency of atoms as ‘the octet rule,’ and it guides us in predicting how atoms combine to form molecules and compounds.
Covalent Bonds and Lewis Diagrams of Simple Molecules
The simplest example to consider is hydrogen (H), which is the smallest element in the periodic table with one proton and one electron. Hydrogen can become stable if it achieves a full valence level like the noble gas that is closest to it in the periodic table, helium (He). These are exceptions to the octet rule because they only require 2 electrons to have a full valence level.
Two H atoms can come together and share each of their electrons to create a ‘covalent bond.’ The shared pair of electrons can be thought of as belonging to either atom, and thus each atom now has two electrons in its valence level, like He. The molecule that results is H2, and it is the most abundant molecule in the universe.
The Lewis formalism used for the H2 molecule is H:H or H—H. The former, known as a ‘Lewis dot diagram,’ indicates a pair of shared electrons between the atomic symbols, while the latter, known as a ‘Lewis structure,’ uses a dash to indicate the pair of shared electrons that form a covalent bond. More complicated molecules are depicted this way as well.
Now consider the case of fluorine (F), which is found in group VII (or 17) of the periodic table. It therefore has 7 valence electrons and only needs 1 more in order to have an octet. One way that this can happen is if two F atoms make a bond, in which each atom provides one electron that can be shared between the two atoms. The resulting molecule that is formed is F2, and its Lewis structure is F—F.
After a bond has formed, each F atom has 6 electrons in its valence level which are not used to form a bond. These non-bonding valence electrons are called ‘lone pairs’ of electrons and should always be indicated in Lewis diagrams.
Procedure for Drawing Simple Lewis Structures
We have looked at how to determine Lewis structures for simple molecules. The procedure is as follows:
- Write a structural diagram of the molecule to clearly show which atom is connected to which (although many possibilities exist, we usually pick the element with the most number of possible bonds to be the central atom).
- Draw Lewis symbols of the individual atoms in the molecule.
- Bring the atoms together in a way that places eight electrons around each atom (or two electrons for H, hydrogen) wherever possible.
- Each pair of shared electrons is a covalent bond which can be represented by a dash.
Multiple bonds can also form between elements when two or three pairs of electrons are shared to produce double or triple bonds, respectively. The Lewis structure for carbon dioxide, CO2, is a good example of this.
In order to achieve an octet for all three atoms in CO2, two pairs of electrons must be shared between the carbon and each oxygen. Since four electrons are involved in each bond, a double covalent bond is formed. You can see that this is how the octet rule is satisfied for all atoms in this case. When a double bond is formed, you still need to show all electrons, so double dashes between the atoms show that four electrons are shared.
Show SourcesBoundless vets and curates high-quality, openly licensed content from around the Internet. This particular resource used the following sources:
http://en.wiktionary.org/wiki/octet_rule
Wiktionary
CC BY-SA 3.0.
Lewis Dot Structure For Covalent Compounds
http://www.chem1.com/acad/webtext/chembond/cb03.html#SEC2
Steve Lower’s Website
CC BY-SA.
http://oer.avu.org/bitstream/handle/123456789/42/Introductory%20Chemistry%201.pdf?sequence=4
GNU FDL.
https://commons.wikimedia.org/wiki/File:Acetic_acid_oxidation_state_analysis.png
Wikimedia
Public domain.
http://commons.wikimedia.org/wiki/File:Miri4.png
Wikimedia Commons
CC BY-SA 3.0.
http://commons.wikimedia.org/wiki/File:Lewis_carbon_dioxide.gif
Wikimedia Commons
Public domain.
http://commons.wikimedia.org/wiki/File:Carbon-dioxide-octet-Lewis-2D.png
Wikimedia Commons
Public domain.
http://commons.wikimedia.org/wiki/File:Covalent.svg
Mikimedia Commons
CC BY-SA 2.5.
Covalent Lewis Dot Structure
https://commons.wikimedia.org/wiki/File:Ikatan_kovalen_pada_molekul_F2.jpg
Wikimedia
CC BY-SA 3.0.
Lewis Dot Structure Covalent Bonds Calculator Worksheet
http://commons.wikimedia.org/wiki/File:Acqua_Lewis.png
Wikimedia Commons
CC BY-SA 3.0.
